Abstract
Introduction: Infection with severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) can result in a cytokine storm that is associated with poor patient outcomes. Despite extensive research, the specific factor associated with SARS-CoV-2 infection that mediates the inflammatory response remains unidentified. We hypothesized that one or more of these proteins is secreted to the bloodstream to induce a systematic inflammatory cytokine response leading to the cytokine storm in patients. We demonstrate that SARS-CoV-2 encoded Open Reading Frame 8 (ORF8) protein is abundantly secreted as a glycoprotein into culture supernatant in vitro and into the bloodstream in patients with COVID-19. Glycosylated ORF8 stimulates monocytes to produce a set of pro-inflammatory cytokines through an NOD-like receptor family pyrin domain-containing 3 (NLPR3)-mediated inflammasome response, which can be inhibited by select NLRP3 inhibitors.
Methods: Following institutional review board approval, serum samples were collected from Mayo Clinic COVID-19 infected patients. Lentiviral constructs expressing SARS-CoV-2 proteins were used to express SARS-CoV-2 proteins in HEK293 cells. Polymerase chain reaction and Luminex multiplex assays were used to detect cytokines induced by COVID-19 proteins. Single-cell RNA sequencing (scRNA-Seq) and flow cytometry were used to identify specific leukocyte populations responsible for cytokine release upon ORF8 stimulation. Immunoblotting, mass spectroscopy, and immunofluorescence microscopy were used to quantify and localize proteins of interest.
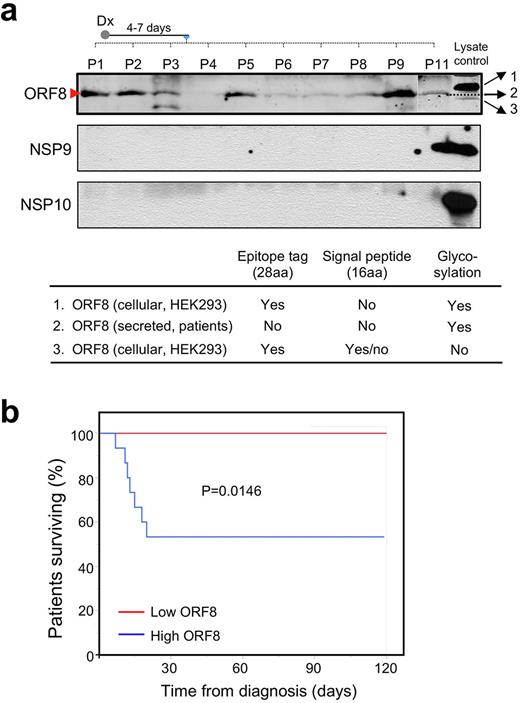
Results: We transduced HEK293 cells with lentiviral constructs that express each SARS-CoV-2 protein and identified ORF8 as abundantly secreted. We treated human peripheral blood mononuclear cells (PBMCs) from healthy donors in vitro with conditioned media (CM) containing major secreted non-structural proteins (NSP) or secreted ORF8 followed by an assessment of cytokine mRNA expression. We found that none of the secreted NSP proteins induced cytokine expression while ORF8-containing CM (ORF8-CM) induced the mRNA expression of IL1b, IL6, IL8, and CCL2 up to 5-fold in PBMCs from select donors. scRNA-Seq on PBMCs treated with ORF8-CM showed that treatment with ORF8 induced IL1b, IL8, and CCL2 expression in CD14, CD16, CD68, and HLA-DR positive activated monocytes.
Flow cytometry analysis of PBMCs stimulated with purified ORF8 showed significant increases of IL1β protein expression only in classical (CD14+/CD16-) and intermediate (CD14+/CD16-) monocyte subsets while no significant changes to cells of other types. scRNA-Seq and Western analyses demonstrated that ORF8 activates NOD-like receptor family pyrin domain-containing 3 (NLPR3) leading to the production of IL1b protein. Furthermore, ORF8-mediated IL1β cytokine induction was abolished in NLPR3 knockdown THP1 cells, demonstrating the NLRP3 dependency of ORF8-mediated cytokine response in human monocytes.
We then examined the correlation between blood ORF8 levels and patients’ survival outcome in 25 patients with COVID-19 infection (Figure 1a). After 120 days of follow-up, all seven fatalities were exclusively associated with the ORF8-high group while all patients in ORF8-low group had minimal morbidity and without any events of death (Figure 1b). Importantly, the ORF8-initiated NLPR3-mediated inflammasome pathway can be effectively inhibited by the NLRP3 inhibitor MCC950 at a nanomolar dose range, demonstrating that targeting ORF8/NLRP3 is a promising strategy for treating patients with symptomatic COVID-19.
Conclusion: We demonstrate that the virus-encoded ORF8 protein is abundantly secreted as a glycoprotein in vitro and in patients with COVID-19. ORF8 specifically targets CD14 + monocytes to produce pro-inflammatory cytokines through NLRP3 mediated inflammasomal activation. Importantly, ORF8 protein level in the blood of patients correlates with severity and disease-specific mortality in with acute SARS-CoV-2 infection. The ORF8-induced inflammasome response was readily inhibited by the NLRP3 inhibitor MCC950 in vitro. Our study identifies a dominant cause of pathogenesis, its underlying mechanism, and a potential new treatment for severe COVID-19 infection.
Disclosures
Ansell:SeaGen: Research Funding; Takeda: Research Funding; Bristol Myers Squibb: Research Funding; Regeneron: Research Funding; Affimed: Research Funding; Pfizer: Research Funding; ADC Therapeutics: Research Funding. Witzig:Karyopharm: Other: Clinical Trail Support; ADC Therapeutics: Membership on an entity's Board of Directors or advisory committees; Curio Science: Honoraria; Kura Oncology: Other: Clinical Trail Support.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal